Research Spotlight: Mass and Charge Balance Model for Predicting Oxide Growth and Dissolution of Corroding Alloys
Alloy 800 pipes
Many alloys have excellent corrosion resistance due to the protective oxide films on their surfaces. However, they all still corrode at certain rates under specific conditions of temperature, pH, the presence of solution impurities, or under radiation. The conditions that an alloy experiences vary greatly depending on the application. For example, an alloy used in a marine environment (cold salty water) experiences a very different environment from an alloy used in the pipes that transport water in a nuclear power plant (high temperatures, pressure, radiation). Predicting the mechanisms and rates of corrosion under a given combination of conditions is very challenging, and is a major part of the ongoing research of the Wren group. The “Mass and Charge Balance” (MCB) model described here is a mathematical model that can predict how corrosion of alloys will progress under a range of different conditions.
Corrosion involves surface redox reactions (metal oxidation coupled with solution reduction) and transfer of the charged species involved in the redox reactions to and from the metal and oxide surfaces. The presence of an oxide film hinders the transfer of the electrons or metal cations, slowing the rate of corrosion. Before we developed the MCB model, some models for predicting corrosion rates in the presence of an oxide film already existed. However, these models did not specifically define the driving force for corrosion as a function of the established electrochemical data for the alloys under study. No existing models took into account changes in oxide composition and layer structure as corrosion progresses. These shortcomings limited the predictive capabilities and the application ranges of these models. The MCB model was designed to overcome some of these limitations.
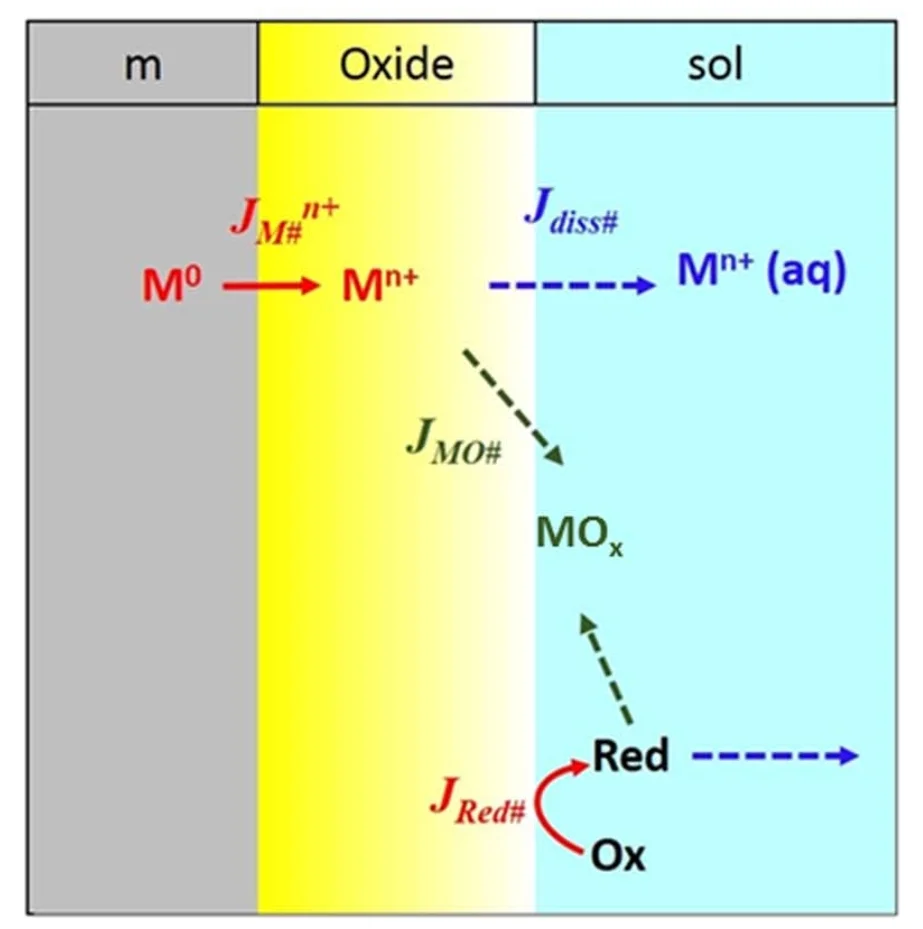
Fluxes considered in the MCB model
The fluxes that the MCB model calculates correspond to measurable quantities: the current, the oxide thickness and the amount of dissolved metal as a function of corrosion time. These are all independently measurable quantities that can be verified experimentally. We have applied the MCB model to simulate the potentiostatic polarization (simulation of particular oxidizing conditions) of a number of alloys including carbon steel, stainless steel, Co-Cr alloy Stellite 6, and FeNi-Cr alloys Inconel 600 and Alloy 800. The modelled results correlated well with experimental data.
The MCB model offers significant advantages over previous models. It can be applied in a range of corrosion scenarios where direct measurements are not possible, allowing for more accurate prediction of the rates of degradation of alloys in varying environments. The MCB model is being further developed to incorporate the rate or flux equations for solid-state oxide conversion processes and dissolution processes such as surface hydration/hydrolysis, and migration/diffusion.
The MCB model considers corrosion to consist of four elements: electrochemical redox reactions at the metal/oxide and oxide/solution interfaces, the transport of charged species across the oxide film, metal oxide formation and growth, and metal ion dissolution. The rates of the individual elementary reactions/processes in the model are formulated using classical chemical reaction rate, and mass and charge flux equations. For example, the oxidation flux is modelled using a modified Butler-Volmer equation that takes into account the potential drop across the oxide layer. These flux equations can be numerically solved using any standard computer software differential equation solver (we used MATLAB). The equations are solved repeatedly in an iterative (stepwise) process, giving the fluxes of mass and charge involved in dissolution of the alloy into solution, and oxide formation, as a function of time.
Current observed during polarization at -0.6 VSCE of (left panel) Co-Cr alloy Stellite 6 and (right panel) Fe-Ni-Cr alloy 800 at (top row) pH 10.6 and (bottom row) pH 8.4. Experimental results are in black and modelling results are in red.
Our publication on this work is:
Mass and Charge Balance (MCB) Model Simulations of Current, Oxide Growth and Dissolution during Corrosion of Co-Cr Alloy Stellite-6 M. Momeni, M. Behazin, and J. C. Wren, J. Electrochem. Soc. 163 (3) C94-C105 (2016)
And it can be found here (you may need to have access through your institution):